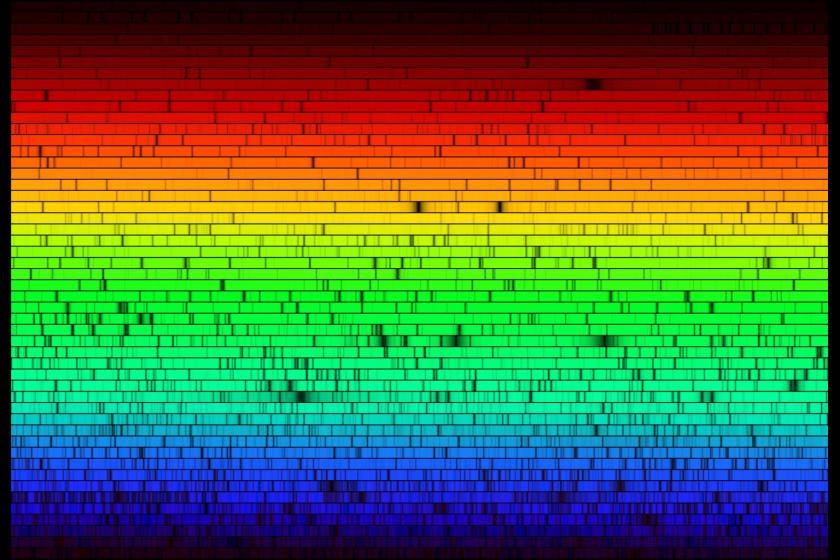
Full Spectrum
By Matt Carroll
Before we even talk about the term “full spectrum“, please take a moment to enjoy the high resolution spectrograph of “white light” from the sun pictured at the top of the article, courtesy of the folks at NOAO.
(They offer even higher resolution versions at the link. They also offer a hi-res spectrograph of the sun’s entire spectrum – white light, plus everything else.)
Note the numerous gaps as we get on with the discussion!
History
The original electric light was an incandescent light bulb.
Incandescent bulbs produce a full spectrum light not unlike the spectrum of white light put out by the sun at a certain time of the day.
In contrast to these old-style lights, electric lights we are familiar with in the hobby such as fluorescent lamps and high intensity discharge (HID) metal halide lamps all put out nearly monochromatic light mostly based on the emission of mercury vapor at various combinations of pressure and temperature. Although sodium lights are another monochromatic alternative.
Here’s an example spectrum of mercury’s color emission from Wikipedia:
Not full spectrum at all. To the eye it’s more of an ugly green light with very few wavelengths of light present.
Let’s look at more examples!
Incandescent
 Here’s a sample of the output from my spectrometer with the spectrum provided by a GE Reveal incandescent bulb.
Here’s a sample of the output from my spectrometer with the spectrum provided by a GE Reveal incandescent bulb.
This is a bulb with a bluish neodymium coating that causes the spectrum to be choppier than a regular clear glass incandescent bulb. That is the reason for the few dim spots in the spectrum.
Fluorescent
Most cheap, household fluorescents aren’t that far removed from the basic mercury emission, which you will recall only puts out a few spikes of color in red green and blue.
 Here’s a sample of the output from my spectrometer with the spectrum provided by an Ikea branded 11-watt compact florescent (CFL) bulb:
Here’s a sample of the output from my spectrometer with the spectrum provided by an Ikea branded 11-watt compact florescent (CFL) bulb:
 Next is a great example of a Phillips florescent bulb that has been heavily modified to be “full spectrum”:
Next is a great example of a Phillips florescent bulb that has been heavily modified to be “full spectrum”:
From comparing those two photos, it’s clear what folks meant when they came up with the term “full spectrum”.
It meant not missing 90% of the color spectrum. Pretty sensible.
(Interestingly, you can still see the mercury emission lines standing out among the rest of the spectrum generated.)
LED
In reefing, “full spectrum” is a different thing altogether. A “full spectrum” LED light does not restore 90% of the color missing from ordinary white LED’s, for example. In fact, the term doesn’t really have any inherent color meaning.
Instead, it is used as a term to contrast with either the light given off by ordinary “white light” LEDs or a basic blend of blue and white light LEDs.
The problem with this is that white LED’s are already very full spectrum – there are merely a few weak wavelengths below the greens and above the blues.
 Here’s a sample of the output from my spectrometer with the spectrum provided by a generic 6500K “white” LED bulb:
Here’s a sample of the output from my spectrometer with the spectrum provided by a generic 6500K “white” LED bulb:
Unlike the CFL picture above, this looks “full spectrum”. Very similar to the incandescent photos. If you have good eyesight or look hard, you can see the dim spot in the spectrum. It is real, but it’s barely there.
Confusingly, in practice, “full spectrum” reef lights do nothing about missing wavelengths.
What they do, generally speaking, is start with a solid, blue+white LED “reef blend” and add RGB (red, green and blue) LED’s so folks can add a decorator effect where one or more of the colors is exaggerated.
How added RGB is supposed to translate into filling that little blue-green wavelength gap in the photo – and why “full spectrum” is the marketing term they chose to use – is anyone’s guess.
(And please don’t tell anyone about the holes in the spectrum of sunlight or it’ll be deemed not full spectrum too!)
Stay tuned to the Internet Reefer’s Decoder Ring series for more!
